Frailty, infections, and bispecific antibodies in multiple myeloma: redefining supportive care in the era of T-cell redirection
Abstract
Multiple myeloma (MM) is a plasma cell malignancy characterized by profound immune dysfunction, predisposing patients to severe infections. Disease- and treatment-related immunosuppression, particularly with proteasome inhibitors, immunomodulatory agents, anti-CD38 antibodies, and bispecific T-cell redirecting antibodies, further exacerbate vulnerability. Frailty, reflecting reduced physiological reserve and cumulative comorbidities, has emerged as a critical determinant of infectious risk, independent of chronological age or apparent clinical fitness. Evidence from ALCYONE and MAIA trials demonstrates that frail patients experience higher rates of severe infections, yet paradoxically, infection rates among seemingly robust patients may approximate those of frail individuals, highlighting limitations in conventional risk stratification. Bispecific T cell redirecting antibodies impose sustained immune perturbations, including B-cell aplasia, hypogammaglobulinemia, and T-cell dysregulation, amplifying infection susceptibility and treatment-related morbidity. Growing clinical and translational evidence indicates that immunoglobulin replacement therapy (IgRT), particularly when administered preemptively, substantially reduces infection rates in patients receiving anti-BCMA bispecific T cell redirecting antibodies. Real-world and mechanistic studies demonstrate that IgRT restores humoral protection and mitigates severe infectious complications, supporting its integration as a central preventive strategy rather than a reactive rescue measure. Integrating frailty assessment with proactive immunologic support represents a precision medicine approach to infection prevention, guiding prophylaxis, monitoring, and early intervention. Future strategies should combine longitudinal frailty evaluation, immune profiling, and disease- and treatment-related factors to optimize individualized infection-risk management in MM patients treated with T-cell–redirecting therapies. Aligning therapeutic innovation with targeted supportive care is essential to maximize efficacy, safety, and durable outcomes in this vulnerable population.
Introduction
Multiple myeloma (MM) is a malignant plasma cell disorder characterized by clonal expansion of antibody-producing cells within the bone marrow, leading to immune dysfunction, bone destruction, cytopenias, and progressive end-organ damage1. It predominantly affects older adults, with a median age at diagnosis close to 70 years, and remains incurable despite major therapeutic advances. From the earliest stages of the disease, MM is associated with profound defects in both humoral and cellular immunity, including suppression of normal immunoglobulin production, impaired antigen presentation, and dysregulated T-cell function2. These alterations translate clinically into a persistently high burden of serious infections across the disease course. Immune dysfunction is further amplified by cumulative exposure to proteasome inhibitors, immunomodulatory drugs, anti-CD38 monoclonal antibodies, and, more recently, highly immunosuppressive cellular and T-cell-redirecting therapies3.
Frailty and Infection Risk in the Era of Bispecific Antibodies
Bispecific T-cell redirecting antibodies have dramatically reshaped the treatment landscape of relapsed and refractory MM, delivering unprecedented response rates in patients with otherwise limited options4. Their mechanism of action, based on continuous redirection of cytotoxic T cells toward plasma cell antigens, explains both their efficacy and their toxicity profile. Bispecific T cell redirecting antibodies simultaneously bind CD3 on T cells and tumor-associated antigens such as B-cell maturation antigen (BCMA) or G-protein coupled receptor family C group 5 member D (GPRC5D) on malignant plasma cells, leading to immune synapse formation, T-cell activation, and target cell lysis. Sustained T-cell activation, prolonged B-cell aplasia, deep and persistent hypogammaglobulinemia, recurrent cytopenias, and cytokine-mediated immune dysregulation collectively result in a state of profound secondary immunodeficiency5. Importantly, toxicity profiles differ according to the target antigen. BCMA-directed bispecific antibodies are characterized by profound and sustained hypogammaglobulinemia due to depletion of normal plasma cells, leading to a high burden of infections, including opportunistic infections. In contrast, GPRC5D-directed bispecific antibodies are associated with a distinct pattern of on-target off-tumor toxicities, including skin rash, nail disorders, and oral toxicities such as dysgeusia and xerostomia, reflecting GPRC5D expression in epithelial tissues, with a comparatively different infectious risk profile (Figure 1). For patients, this biological vulnerability translates into frequent and severe infections, which have emerged as one of the leading causes of treatment interruption, hospitalization, and early mortality during bispecific T cell redirecting antibodies therapy5.
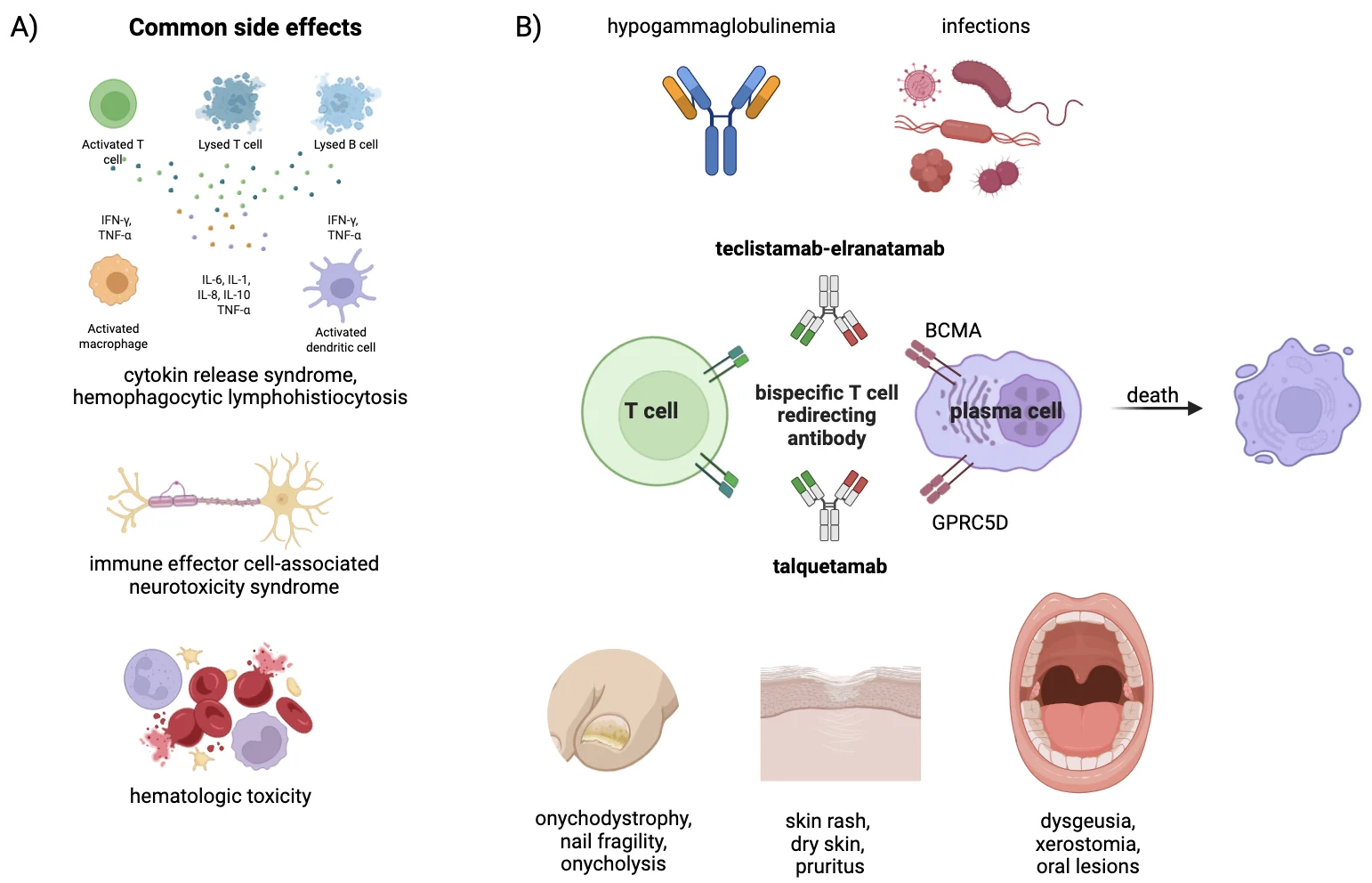
Figure 1. Common adverse events during bispecific T cell redirecting antibody treatment and their mechanism of action. A) common side effects during bispecific T cell redirecting antibodies therapy. B) bispecific T cell redirecting antibody mechanism of action and main related toxicities.
As bispecific T cell redirecting antibodies move from late-line salvage settings into earlier disease phases and broader patient populations, infection-risk stratification based solely on traditional factors is increasingly inadequate. Neutropenia, disease stage, and number of prior therapies capture treatment exposure but fail to reflect the host’s ability to tolerate sustained immune stress. In this context, frailty provides a unifying framework that goes beyond conventional risk factors6. Unlike age or comorbidity counts, frailty integrates biological aging, functional impairment, and loss of physiological reserve, all of which are closely linked to immune senescence6. In MM, frailty has consistently predicted inferior survival, higher rates of treatment discontinuation, and excess non-hematologic toxicity, yet its role in shaping infectious risk has only recently been explored in depth.
Data from large randomized trials in non-transplant-eligible newly diagnosed MM confirm that frail patients experience substantially higher rates of severe infections than fitter individuals7. However, analyses from the ALCYONE and MAIA trials reveal that the relationship between frailty and infection is not strictly linear8. In ALCYONE, Mateos et al. showed that patients categorized as fit did not consistently experience fewer infections than those classified as intermediate, challenging the assumption that apparent robustness equates to lower infectious risk9. Similar observations emerged from the MAIA frailty analysis reported by Facon et al10. Together, these findings highlight the heterogeneity concealed within broad frailty categories and emphasize that current frailty tools, designed to predict survival and treatment tolerance, lack precision for identifying patients at highest infectious risk. Disease burden, treatment intensity, and immune reserve likely interact with frailty in ways not captured by existing models.
When frailty intersects with bispecific T cell redirecting antibodies therapy, the clinical consequences become particularly pronounced. Bispecific T cell redirecting antibodies impose a sustained immunosuppressive pressure that challenges even preserved immune systems4. In patients with reduced physiological and immunological reserve, this additional burden may push host defenses beyond a critical threshold. Functional impairment can delay recognition of early infectious symptoms, while limited reserve reduces tolerance to systemic inflammatory responses and sepsis11. Clinically, this results in a subgroup of patients at exceptionally high risk of high-grade infections, prolonged hospitalizations, and early treatment discontinuation, ultimately undermining the benefit of these otherwise transformative therapies12.
Preemptive Immune Support and Precision Supportive Care
This convergence calls for a reappraisal of supportive care in the era of T-cell redirection. Among preventive strategies, immunoglobulin replacement therapy (IgRT) stands out as both biologically rational and increasingly evidence based13. Secondary hypogammaglobulinemia is common in advanced MM and is further exacerbated by bispecific T cell redirecting antibodies through sustained depletion of normal plasma cells14. Low polyclonal IgG levels are strongly associated with recurrent and severe infections, yet immunologic monitoring and IgRT remain inconsistently applied in routine practice. Importantly, reliance on fixed IgG thresholds alone may be insufficient to guide IgRT initiation, as functional humoral impairment and patient-specific factors such as frailty, prior infections, and treatment-related immune depletion may significantly influence infection risk (Figure 1).
Recent real-world and translational studies provide compelling evidence that immunoglobulin supplementation can meaningfully reduce infectious morbidity in patients treated with anti-BCMA bispecific T cell redirecting antibodies. In a multi-institutional cohort of teclistamab-treated patients, Cheruvalath et al. reported a marked reduction in infections among those receiving preemptive intravenous IgRT compared with those who did not15. Frerichs et al. demonstrated that teclistamab profoundly impairs humoral immunity and that IgRT partially restores protective antibody levels, translating into fewer infections16. These findings are reinforced by large real-world studies showing a dramatic reduction in serious infections associated with IVIg use in patients treated with anti-BCMA bispecific T cell redirecting antibodies17,18.
From a practical standpoint, IgRT use should consider IgG levels (<400-500 mg/dL, depending on clinical context), infection history, frailty, and treatment intensity. Serum IgG levels represent a practical surrogate marker to support IgRT initiation, although they should not be used in isolation. Early initiation, preferably at bispecific antibodies start or rapid IgG decline, appears more effective than delayed use. Both IVIg and SCIg are options, with dosing tailored to clinical response and IgG levels. Regular monitoring of serum IgG during treatment may help identify patients at increased risk of severe infections and support timely prophylactic intervention. Key practical recommendations for infection prevention, immunoglobulin monitoring, and supportive care during bispecific antibody therapy are summarized below (Table 1)4.
Table 1. Key International Myeloma Working Group (IMWG) recommendations for the optimal use of bispecific T cell redirecting antibodies in multiple myeloma.
| Clinical domain | Key IMWG recommendations |
|---|---|
| Patient selection | Careful baseline assessment of frailty, performance status, prior infections, disease burden, and organ function before treatment initiation. |
| Treatment initiation | Step-up dosing and premedication are recommended to reduce CRS risk. |
| CRS monitoring and management | Close monitoring during step-up doses and early cycles; prompt supportive care and early tocilizumab for persistent or symptomatic CRS. |
| Neurotoxicity surveillance | Baseline neurological assessment and serial monitoring for ICANS, especially during treatment initiation. |
| Hematologic toxicity management | Regular blood count monitoring; use growth factors, transfusions, and dose delays as needed for cytopenias. |
| Infection prevention: Antimicrobial prevention | Vaccination, antiviral prophylaxis (HSV/VZV), Pneumocystis prophylaxis, and antibacterial/antifungal prophylaxis according to risk. |
| Infection prevention: Immunoglobulin monitoring | Regular serum immunoglobulin monitoring throughout therapy. |
| Infection prevention: IgRT use | IgRT should be considered in patients with severe hypogammaglobulinemia (especially IgG <400 mg/dL), recurrent or severe infections. |
| Target-specific considerations | BCMA-directed antibodies carry higher risk of prolonged hypogammaglobulinemia and infections; GPRC5D-directed antibodies require additional monitoring for skin, nail, and oral toxicities. |
| Longitudinal follow-up | Dynamic reassessment of frailty, immune status, and infectious complications throughout treatment and after discontinuation. |
Importantly, emerging data suggest that timing matters. Preemptive IgRT, rather than reactive use after recurrent infections, appears to confer the greatest benefit, supporting a prevention-oriented approach based on anticipated risk. Piron et al. showed that systematic IgRT prophylaxis significantly reduced infections in patients receiving anti-BCMA bispecific T cell redirecting antibodies, supporting a shift toward prevention-oriented strategies19. Similar results were observed in contemporary teclistamab cohorts managed with routine IVIg prophylaxis20. In this context, IgRT should no longer be viewed as a rescue therapy but as a core component of preventive care, particularly for biologically vulnerable patients. Benefit extending beyond those with profoundly low IgG levels further suggests that static thresholds underestimate true immunological risk, especially in frail individuals with qualitative immune impairment.
Integrating frailty assessment with proactive immune support represents a practical step toward precision supportive care. Frailty evaluation should inform not only treatment intensity but also anticipatory prophylaxis, monitoring, and early intervention strategies in patients considered for bispecific T cell redirecting antibody therapy (Figure 2). Patients who appear relatively robust may still harbor substantial infectious vulnerability, underscoring the need for repeated and dynamic assessment over time.
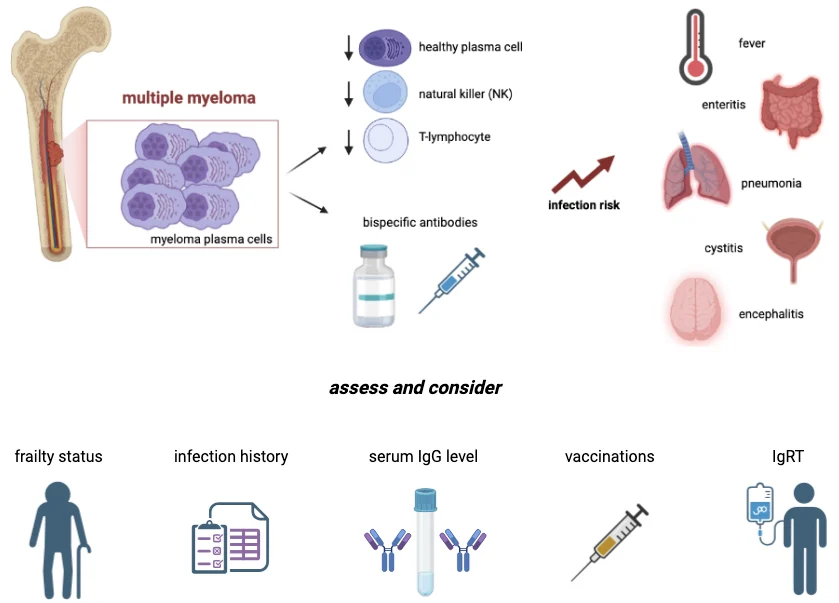
Figure 2. Practical framework for infection risk assessment in multiple myeloma patients receiving bispecific T cell redirecting antibodies. Pathophysiology of immunodeficiency in multiple myeloma and consideration about risk assessment and prevention measures with vaccination and immunoglobulin replacement therapy (IgRT).
Conclusion
Looking ahead, infection-risk stratification must evolve toward integrated models combining frailty, immune profiling, disease burden, and treatment characteristics. Longitudinal assessment of immunoglobulin levels, lymphocyte subsets, and functional status could better identify patients at risk than any single parameter alone. Prospective registries and trials incorporating frailty-based stratification and standardized infection endpoints are urgently needed.
Bispecific T cell redirecting antibodies have transformed the therapeutic horizon of MM, but they have also exposed the limits of conventional supportive care. Frailty should be recognized as a dynamic and actionable determinant of infectious risk. Aligning innovative anti-myeloma therapies with equally proactive preventive strategies, including early IgRT, is essential to translate biological efficacy into durable clinical benefit. In the era of T-cell redirection, protecting the patient is no longer ancillary to treating the tumor; it is a prerequisite for therapeutic success.
Conflict of interest statement: A.G.S. has received speaker honoraria from Sanofi, Amgen, and AstraZeneca; has participated in advisory boards for Pfizer and Menarini; and received travel support for educational purposes from Janssen-Cilag. A.G.S. received also research support from Pfizer. C. C. has received speaker honoraria from Janssen, Bristol Myers Squibb/Celgene, AbbVie, Pfizer, Takeda, Amgen, Sanofi/Aventis, GlaxoSmithKline, BeiGene, Stemline Therapeutics, Menarini, Karyopharm Therapeutics, and Servier; has participated in advisory boards for Amgen, Janssen, Bristol Myers Squibb/Celgene, AbbVie, Takeda, Pfizer, Sanofi, GlaxoSmithKline, BeiGene, Menarini, Karyopharm Therapeutics, Servier, and Stemline Therapeutics; and has been part of speakers’ bureaus for Amgen, BeiGene, Bristol Myers Squibb/Celgene, GlaxoSmithKline, Janssen, AbbVie, Takeda, Pfizer, Menarini, Servier, Stemline Therapeutics, Karyopharm Therapeutics, and Sanofi/Aventis. R.R. received honoraria and participated in advisory boards for Pfizer, Sanofi, Bristol Myers Squibb-Celgene, Octapharma, Takeda, Janssen-Cilag, AstraZeneca, Menarini, CSL-Behring, and Amgen. A.V. received speaker honorariums from Pfizer, Sanofi, Bristol Myers Squibb, Takeda, Janssen Cilag, AstraZeneca, Menarini, and Amgen. Their potential conflicts of interest do not imply bias or influence on the authors’ opinions or actions. The authors recognise the importance of transparency in the scientific field and are committed to upholding their integrity and maintaining trust in them within the scientific community. The remaining authors declare no competing financial interests.
Funding: This work was funded by “The Italian network of excellence for advanced diagnosis (INNOVA), Ministero della Salute (code PNC-E3-2022-23683266 PNC-HLS-DA) to V.D. and A.G.S.; Complementary National Plan PNC-I.1 “Research initiatives for innovative technologies and pathways in the health and welfare sector” D.D. 931 of 06/06/2022, DARE – DigitAl lifelong pRevEntion initiative (code PNC0000002) to A.V. and R.R.; PNRR-MCNT1-2023-12377893 DEMMMO- “Deciphering multiple myeloma using multiomic approaches for immunotherapy modeling“ (CUP: H93C23001080006) to V.D. and A.G.S. A.G.S. was also funded by Pfizer Global Independent Research and Knowledge Gap Grants.
Ethics approval and consent to participate: Not applicable.
License
© Author (s), [2026].
This is an open access article distributed under the terms of the Creative Commons Attribution 4.0 International License (CC BY 4.0), which permits unrestricted use, distribution, and reproduction in any medium, and unrestricted adaptation and reuse, including for commercial purposes, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
To view a copy of this license, visit https://creativecommons.org/licenses/by/4.0/.
References
-
Kyle RA, Rajkumar SV. Multiple myeloma. Blood. 2008 Mar 15;111:2962-72.
-
Allegra A, Tonacci A, Musolino C, et al. Secondary immunodeficiency in hematological malignancies: focus on multiple myeloma and chronic lymphocytic leukemia. Front Immunol. 2021 Oct 25;12:738915.
-
Jolles S, Giralt S, Kerre T, et al. Agents contributing to secondary immunodeficiency development in patients with multiple myeloma, chronic lymphocytic leukemia and non-Hodgkin lymphoma: a systematic literature review. Front Oncol. 2023 Feb 7;13:1098326.
-
Rodriguez-Otero P, Usmani S, Cohen AD, et al. International Myeloma Working Group immunotherapy committee consensus guidelines and recommendations for optimal use of T-cell-engaging bispecific antibodies in multiple myeloma. Lancet Oncol. 2024 May;25:e205-e216.
-
Ludwig H, Terpos E, van de Donk N, et al. Prevention and management of adverse events during treatment with bispecific antibodies and CAR T cells in multiple myeloma: a consensus report of the European Myeloma Network. Lancet Oncol. 2023 Jun;24:e255-e269.
-
Palumbo A, Bringhen S, Mateos MV, et al. Geriatric assessment predicts survival and toxicities in elderly myeloma patients: an International Myeloma Working Group report. Blood. 2015 Mar 26;125:2068-74.
-
Zhang Y, Liang X, Xu W, et al. Individualized dynamic frailty-tailored therapy (DynaFiT) in elderly patients with newly diagnosed multiple myeloma: a prospective study. J Hematol Oncol. 2024 Jun 24;17:48.
-
Spataro F, Armentaro G, Di Gioia G, et al. Impact of frailty on infection risk in non-transplant eligible multiple myeloma patients: a systematic review and meta-analysis. Leukemia. 2026 Feb 17.
-
Mateos MV, Dimopoulos MA, Cavo M, et al. Daratumumab plus bortezomib, melphalan, and prednisone versus bortezomib, melphalan, and prednisone in transplant-ineligible newly diagnosed multiple myeloma: frailty subgroup analysis of ALCYONE. Clin Lymphoma Myeloma Leuk. 2021 Nov;21:785-798.
-
Facon T, Cook G, Usmani SZ, et al. Daratumumab plus lenalidomide and dexamethasone in transplant-ineligible newly diagnosed multiple myeloma: frailty subgroup analysis of MAIA. Leukemia. 2022 Apr;36:1066-1077.
-
Li H, Manwani B, Leng SX. Frailty, inflammation, and immunity. Aging Dis. 2011 Dec;2:466-73.
-
Vacca A, Melaccio A, Sportelli A, et al. Subcutaneous immunoglobulins in patients with multiple myeloma and secondary hypogammaglobulinemia: a randomized trial. Clin Immunol. 2018 Jun;191:110-115.
-
Jolles S, Michallet M, Agostini C, et al. Treating secondary antibody deficiency in patients with haematological malignancy: European expert consensus. Eur J Haematol. 2021 Apr;106:439-449.
-
Raje N, Anderson K, Einsele H, et al. Monitoring, prophylaxis, and treatment of infections in patients with MM receiving bispecific antibody therapy: consensus recommendations from an expert panel. Blood Cancer J. 2023 Aug 1;13:116.
-
Cheruvalath H, Clennon A, Shresth A, et al. Effects of intravenous immunoglobulin supplementation (IVIG) on infections in recipients of teclistamab therapy for multiple myeloma (MM): a multi-institutional study. Blood. 2024;144 Suppl 1:256.
-
Frerichs KA, Verkleij CPM, Mateos MV, et al. Teclistamab impairs humoral immunity in patients with heavily pretreated myeloma: importance of immunoglobulin supplementation. Blood Adv. 2024 Jan 9;8:194-206.
-
Lancman G, Parsa K, Kotlarz K, et al. IVIg use associated with ten-fold reduction of serious infections in multiple myeloma patients treated with anti-BCMA bispecific antibodies. Blood Cancer Discov. 2023 Nov 1;4:440-451.
-
Mohan M, Monge J, Shah N, et al. Teclistamab in relapsed refractory multiple myeloma: multi-institutional real-world study. Blood Cancer J. 2024 Mar 5;14:35.
-
Piron B, Rodrigo L, Tessoulin B, et al. Preemptive immunoglobulin prophylaxis reduces infections in patients treated with anti-BCMA bispecific antibodies. Clin Lymphoma Myeloma Leuk. 2025 Oct 31:S2152-2650(25)04264-8.
-
Sheu M, Molina Garcia S, Patel M, et al. Infection prophylaxis with intravenous immunoglobulin in multiple myeloma patients treated with teclistamab. Oncology. 2025 Oct 23:1-5.





