Outcome of Hematopoietic Stem Cell Transplantation in Pediatric Patients with Severe Aplastic Anemia and Multi-Drug Resistant Bacterial Infections – A Case Series
Abstract
Background: Severe aplastic anemia (SAA) is a life-threatening bone marrow failure syndrome requiring hematopoietic stem cell transplantation (HSCT) for cure. The presence of multidrug-resistant organisms (MDROs) in pediatric patients poses a significant challenge, increasing transplant-related morbidity and mortality. Evidence guiding the timing and feasibility of HSCT in this high-risk population remains limited.
Methods: We retrospectively reviewed four pediatric SAA patients with concurrent MDRO infections who underwent HSCT at a tertiary transplant center. Clinical data, infectious profiles, conditioning regimens, engraftment kinetics, and outcomes were analyzed to identify key determinants of transplant success.
Results: Three of four patients survived beyond one-year post-HSCT with full hematopoietic recovery and sustained donor chimerism (>97%). Infections included extended-spectrum β-lactamase (E. coli, Klebsiella pneumoniae), methicillin-resistant Staphylococcus aureus, vancomycin-resistant Enterococcus, and Stenotrophomonas maltophilia. Tailored antimicrobial therapy guided by susceptibility testing, strict infection control measures, and close collaboration between transplant and infectious disease teams were critical to achieving favorable outcomes. Neutrophil engraftment occurred at a median of 15 days. One patient succumbed to carbapenem-resistant K. pneumoniae sepsis despite maximal therapy.
Conclusion: HSCT can be safely and successfully performed in pediatric SAA patients with active or recent MDRO infections when supported by multidisciplinary management, individualized antimicrobial strategies, and rigorous infection control. Early engraftment remains the key determinant of immune recovery and infection resolution. This case series underscores that MDRO infection should not preclude timely transplantation when appropriate infection stabilization is achieved.
Introduction
Severe aplastic anemia (SAA) is a rare and life-threatening hematologic disorder characterized by hypocellular bone marrow and peripheral pancytopenia. In children, the disease is often idiopathic but may result from inherited bone marrow failure syndromes, drugs, viral infections, or immune-mediated destruction of hematopoietic progenitors. Without definitive treatment, SAA is associated with a high risk of infection and bleeding due to profound marrow failure1,4,5. Hematopoietic stem cell transplantation (HSCT) remains the only curative therapy for pediatric SAA, particularly in patients with matched sibling or unrelated donors. Over the past decades, improved conditioning regimens, infection control measures, and supportive care have significantly enhanced HSCT outcomes3,7. However, the emergence of multidrug-resistant organisms (MDROs) has introduced new challenges in the management of these immunocompromised patients.
MDROs, such as methicillin-resistant Staphylococcus aureus (MRSA), vancomycin-resistant Enterococcus (VRE), and carbapenem-resistant Enterobacteriaceae (CRE) – have become increasingly prevalent among pediatric patients with prolonged hospitalizations, indwelling catheters, and prior exposure to broad-spectrum antibiotics2,9. Infections or colonization with these pathogens before HSCT are associated with higher morbidity and transplant-related mortality3. Nevertheless, delaying transplantation for infection eradication may not be feasible in SAA due to the high risk of life-threatening cytopenias.
Although isolated reports and small series describe HSCT in patients colonized or infected with MDROs, there remains limited evidence on how to optimally time transplantation, select conditioning regimens, and manage peri-transplant infections in this setting. Multidisciplinary coordination among transplant physicians, infectious disease specialists, and critical care teams is critical to optimizing outcomes.
This report describes a series of four pediatric patients with SAA and documented MDRO infections who successfully underwent HSCT at a tertiary care center. We highlight our institutional approach to infection control, antimicrobial management, conditioning strategies, and post-transplant outcomes, aiming to contribute practical insights to this challenging clinical scenario.
Case 1
A previously healthy 6 years old Saudi female with negative family history and negative bone marrow failure (BMF) panel, was diagnosed with SAA. The pre-transplant course was complicated with E coli ESBL liver abscesses, in addition to EBV and CMV viremia, HSV gingivostomatitis, and presumed invasive pulmonary fungal infection. She underwent HSCT after failing horse Anti Thymoglobin as a first line immunosuppressive therapy for SAA, while being on broad spectrum antimicrobials (details of clinical course and antimicrobial therapy in Figure.1). The conditioning regimen included Cyclophosphamide, Fludarabine and Alemtuzumab with calcineurin inhibitor as GVHD prophylaxis; she received a total of 5.46 *10^6 per kg of stem cell from a MUD. Transplant course was complicated with engraftment syndrome in the form of rash and respiratory distress, managed successfully with steroids, in addition to febrile neutropenia, Staphylococcus epidermidis bacteremia, HSV gingivostomatitis, reactivation of CMV viremia and persistent liver abscesses. Neutrophil and platelet engraftment occurred on days +14 and +30, respectively, and the patient was discharged on day +31. Broad-spectrum antimicrobial therapy was continued based on susceptibility profiles, through the neutropenic phase until 5 months post-transplant. Chimerism was persistently more than 96% on days 30, 60, 100, 180, and at 1- and 2-years post-transplant. Currently, she is over 2 years post transplantation, received her post-transplant vaccines and is off all medications, with good outcome.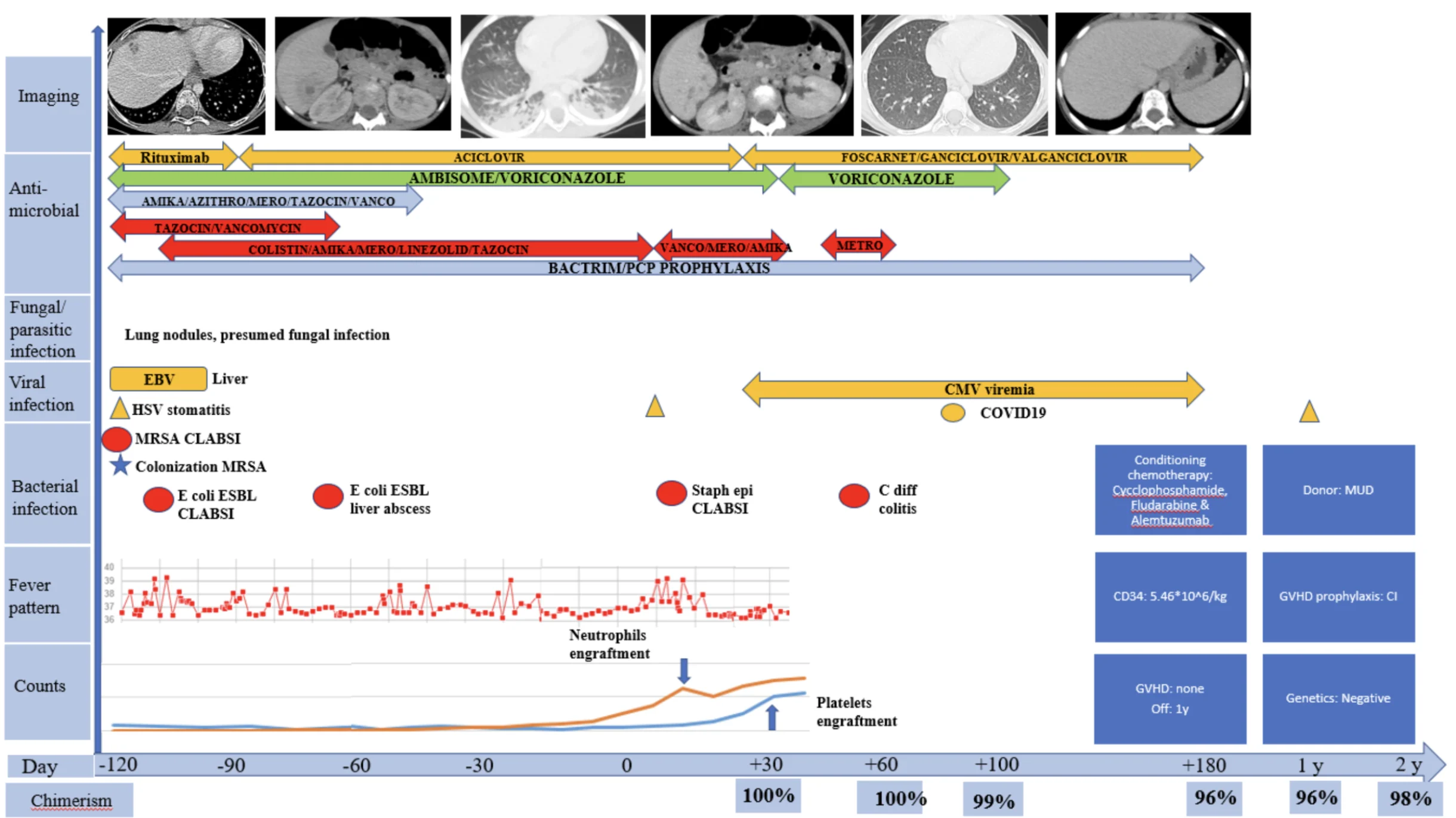
Figure 1; Case 1: Timeline of clinical course, infectious complications, antimicrobial therapy, and hematologic recovery from four months prior to HSCT until 2-years post-transplant.
Case 2
A 10-year-old Saudi female with neurofibromatosis type 1 (NF1), with negative family history of AA and negative BMF panel was diagnosed with SAA. The pre-transplant course was complicated with refractory pediculosis capitis, multiple catheter-related blood stream infections (CLABSI) with various resistant organisms, including: methicillin-resistant Staphylococcus aureus (MRSA), Vancomycin-resistant Enterococcus faecium (VRE), and Escherichia coli ESBL (Figure.2) prolonged febrile neutropenia with ESBL neutropenic colitis and perianal abscesses, MRSA necrotizing lymphadenitis, necrotizing pneumonia, multiple liver hypodense lesions as well as colonization with multi-drug resistant bacteria, in addition to herpetic stomatitis. She received prolonged courses of antimicrobials as detailed in Figure.2 and at 8 months after presentation, she underwent HSCT. She received 5.4*10^6 per kg of stem cells from a MUD. Conditioning regimen included Cyclophosphamide, Fludarabine and ATG with calcineurin inhibitor and MMF as GVHD prophylaxis. The transplant course was complicated with Stenotrophomonas maltophilia CLABSI on day +6, in addition to multiple viral reactions: Parvovirus, CMV and EBV viremia. Engraftment was successfully achieved for both platelets and neutrophils on day +15 and the patient was discharged on day +29. Tailored antimicrobial therapy was continued until day +90 with: Levofloxacin, Linezolid, Metronidazole, Posaconazole and Valganciclovir. Chimerism was persistently more than 97% on days 30, 60, 100, 180, and at 1- and 2-years post-transplant. Currently, she is over 2 years post transplantation, received her post-transplant vaccines and is off all medications, with good outcome.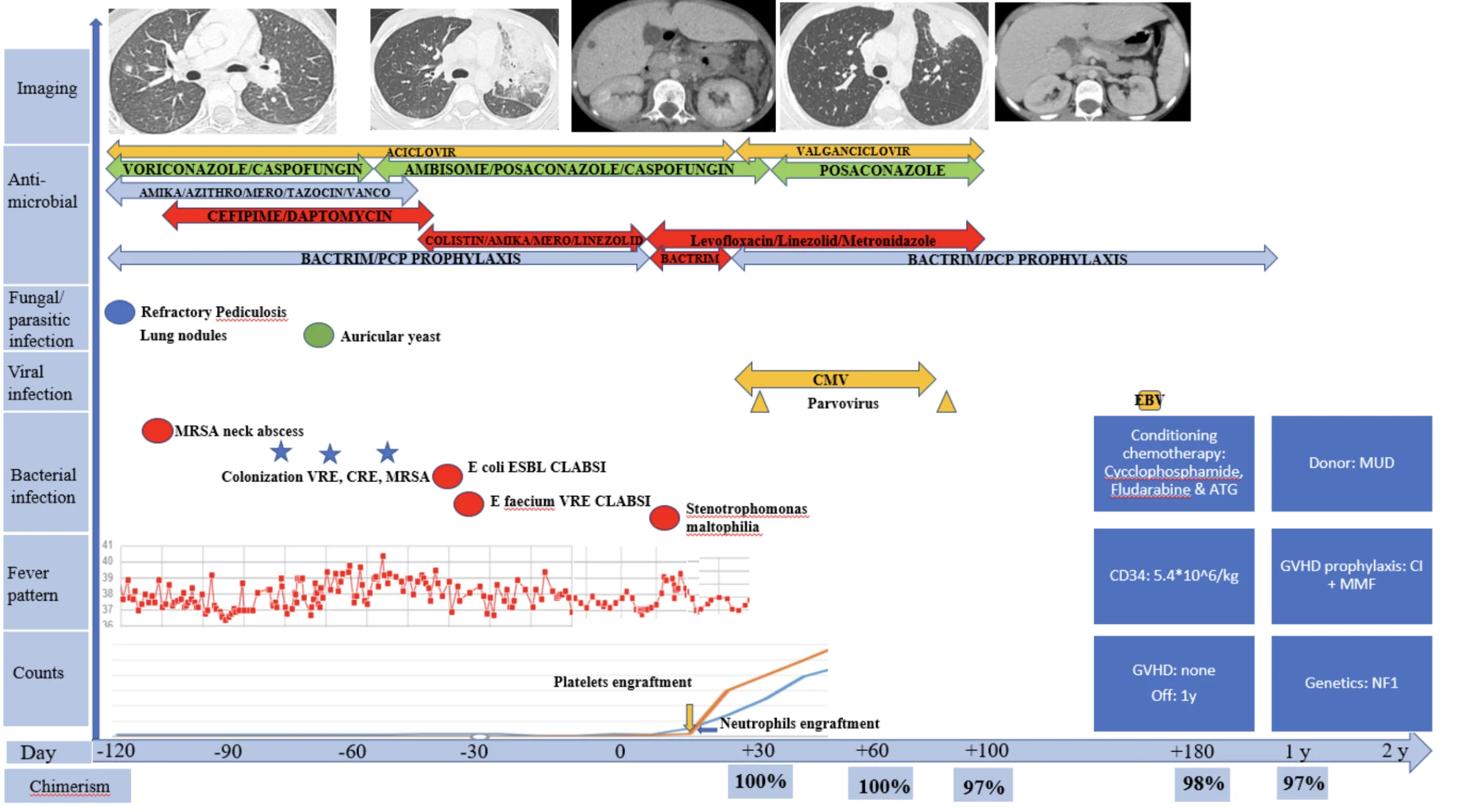
Figure 2; Case 2: Timeline of clinical course, infectious complications, antimicrobial therapy, and hematologic recovery from four months prior to HSCT until 2-years post-transplant.
Case 3
A 3-year-old Saudi female with a family history of a similar condition in her older sibling, who presented with pancytopenia and was diagnosed with SAA. The older sibling passed away at the age of 5 years secondary to SAA prior to transplant; investigations of the deceased sibling revealed a homozygous mutation for TERC gene compatible with the diagnosis of dyskeratosis congenita. Our patient was extensively worked up and had on WES (Whole Exome Sequencing) the same TERC mutation reported in the deceased sibling. Prior to presenting to our institution, she had multiple intensive care admissions for severe pneumonias in an outside hospital. The pre-transplant course was complicated with CLABSI (E. coli), Pneumocystis jiroveci pneumonia (confirmed by lung biopsy) complicated by a residual large pulmonary cyst, adenovirus and EBV viremia, in addition to colonization with MRSA and Klebsiella pneumonia CRE, as well as Streptococcus pneumoniae in nasal PCR. Details of antimicrobials and clinical course are summarized in Figure.3. Transplant was started while being on voriconazole, cefepime and Bactrim. Conditioning chemotherapy included Melphalan, Fludarabine and ATG, and GVHD prophylaxis was with calcineurin inhibitor and MMF. A total of 3.3 *10^6 per kg of stem cells was infused from a MUD. Initially, the HSCT course was smooth until day +2, where she developed fever with epigastric abdominal pain, and was managed with empiric antimicrobials and clinically stabilized. On day +10, the epigastric pain recurred without fever. On day +12, neutrophils engrafted and this was coupled with clinical improvement until day +21, where she started having fever and vomiting, that became intractable on day +24, associated with diarrhea and hypotension, hence she was shifted to PICU with clinical suspicion of gastrointestinal acute GVHD. On day +26, the patient developed bloody diarrhea and was started on inotropic support, on day +29, she underwent sigmoidoscopy and lung biopsy, where the tissue culture and blood culture taken on the same day grew carbapenem-resistant Klebsiella pneumoniae. Unfortunately, on day +31 she passed away, despite the maximal supportive measures, with septic shock.
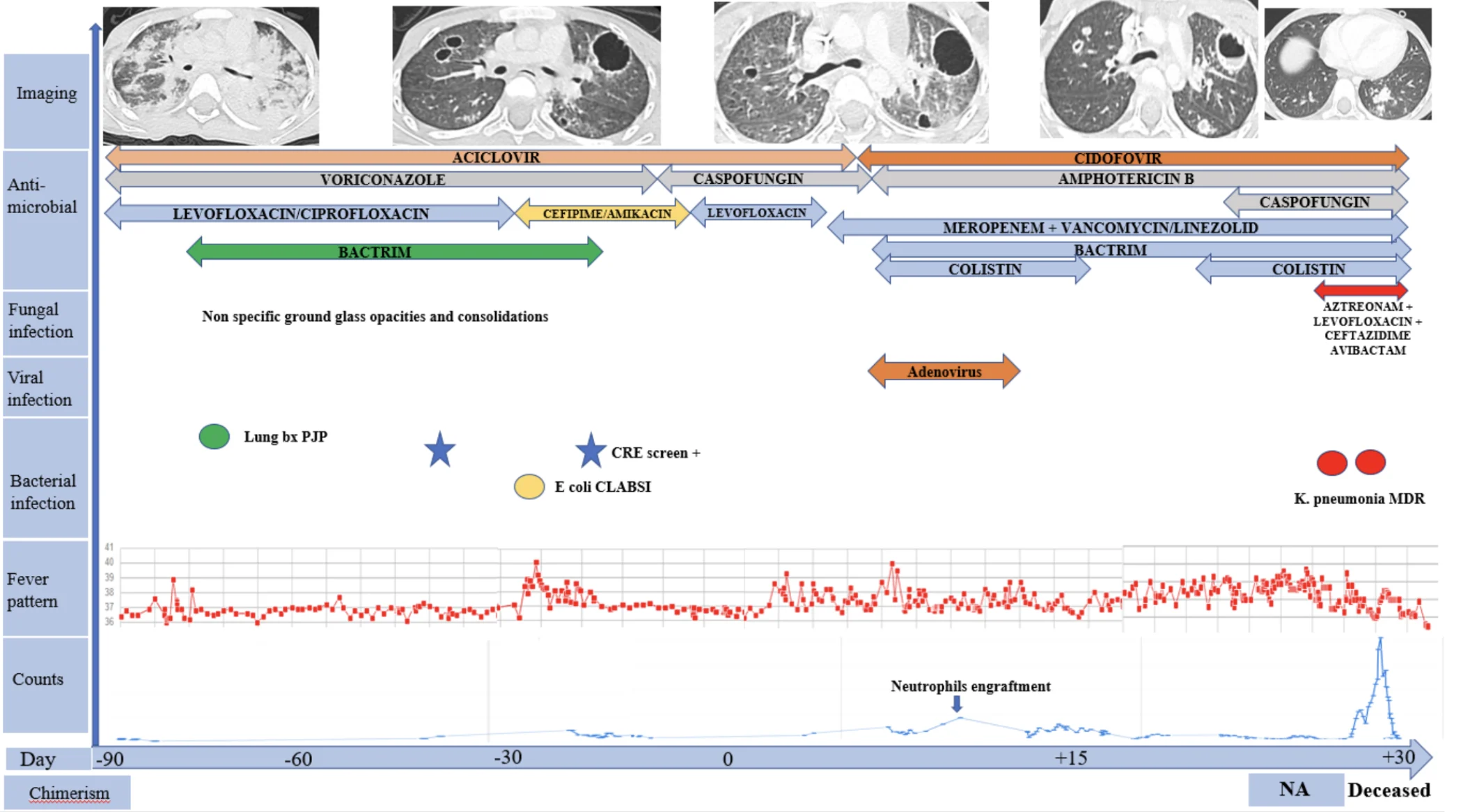
Figure 3; Case 3: Timeline of clinical course, infectious complications, antimicrobial therapy, and hematologic recovery from three months prior to HSCT until the patient’s death on day +31 post-transplant.
Case 4
A 3 years old Yemeni male with negative family history of SAA and negative BMF panel was diagnosed with SAA. The pre-transplant course was complicated with hepatic abscess, thigh abscess, submandibular large draining lymphadenopathy with growth of Stenotrophomonas maltophilia from blood and thigh abscess drained fluid. In addition, he had carbapenem-resistant E. coli bacteremia, and ground glass opacities on chest CT scans concerning for possible invasive fungal infection. Details of antimicrobial therapy and clinical course is detailed in Figure.4. He underwent HSCT after clinical stability while being on Ceftazidime/Avibactam and Levofloxacin for the two main resistant pathogens isolated from the abscesses. Conditioning regimen included Cyclophosphamide, Fludarabine and ATG with calcineurin inhibitor as GVHD prophylaxis. A total of 6*10^6 per kg of stem cells from his fully matched sibling donor were infused. During pre-engraftment phase, the submandibular abscess ruptured spontaneously and grew VRE, he developed Staphylococcus epidermidis bacteremia and the hip abscess continued to drain with negative growth on cultures. In addition, CMV was reactivated. Neutrophils and platelets engrafted on days +20 and +35 respectively. Coverage with broad spectrum antimicrobial coverage tailored for the susceptibility profiles was continued until day +104. Chimerism was persistently more than 97% on days 30, 60, 100, 180, and at 1-year post-transplant. Currently, he is over 18 months post transplantation, he continues to receive post-transplant vaccines, and is off all medications with complete resolution of all previous abscesses clinically and on follow up images.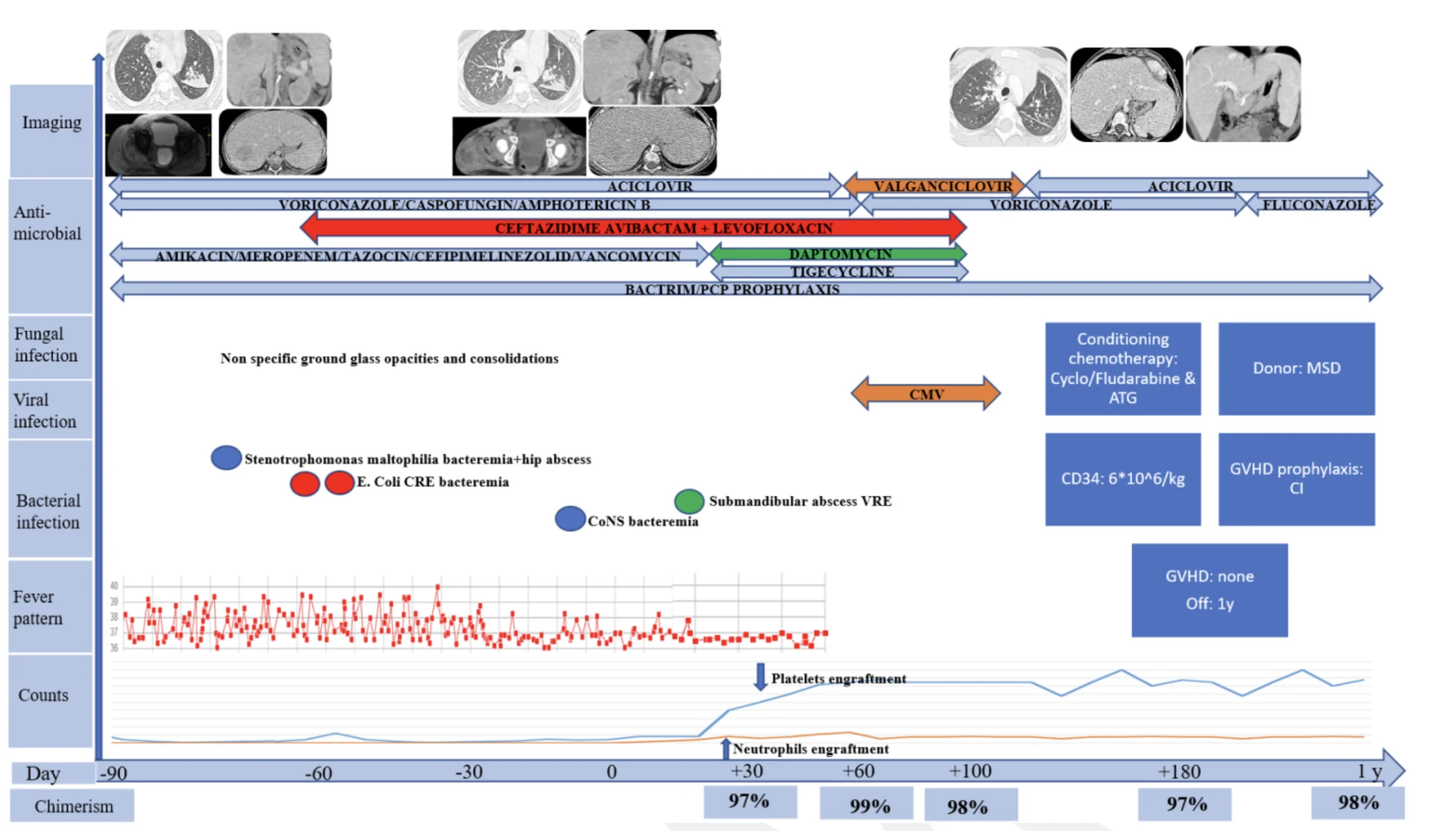
Figure 4: Case 4: Timeline of clinical course, infectious complications, antimicrobial therapy, and hematologic recovery from three months prior to HSCT until 1-year post-transplant.
Discussion
The outcomes of our pediatric patients with SAA and MDRO infections who underwent HSCT are in line with, or even slightly better than, those reported in the literature6,7,8. The MDRO organisms encountered in our pediatric SAA cases mirror those reported in hematopoietic transplant populations. Most infections were caused by resistant Gram-negative bacilli. Literature from pediatric HSCT cohorts shows Klebsiella pneumoniae (often carbapenemase-producing) and Pseudomonas aeruginosa (often multidrug-resistant) among the most common bloodstream isolates, together with occasional VRE and other MDROs1,8,9. In our series, deep-seated infections underscore the breadth of resistant pathogens that can complicate SAA. The colonization or infection with MDROs has been linked to higher non-relapse mortality after HSCT. For example, one study found 5-year survival was only ~43% in MDRO-colonized HSCT patients vs ~66% in non-colonized controls10. Our cases add to the growing evidence that carbapenem-resistant gram-negatives in particular pose a grave risk, but also that successful outcomes are attainable with aggressive management.
Antimicrobials use prior to and throughout transplant was tailored based on each patient’s antibiogram and according to international guidelines. Strict infectious preventive measures, including contact isolation, limitation of visitors, presence of a single watcher throughout the transplant period, in addition to frequent screening for CRE, VRE and MRSA was carried on all of our patients. This served as an important strategy in empiric therapy guidance, where the presence of MDRO colonization allowed us to preemptively broaden empiric coverage during febrile neutropenia. Literature review for similar patients, resulted in viewing diverse cohorts of patients, predominantly adults, with documented MDRO infections1,2,3. Rigorous infection control strategies varied widely and included stringent infection control measures, prophylactic antibiotics, and tailored antimicrobial therapies based on sensitivity patterns.
In our patients, HSCT was initiated as soon as infections were stabilized under therapy, reflecting a case-by-case approach. We tended to use non-myeloablative conditioning chemotherapy, similar to what is reported in the international literature4,5,6. Those included agents such as cyclophosphamide, fludarabine, melphalan and ATG. The choice of regimen was influenced by patient age, comorbidities, and MDRO status. Donor selection criteria emphasized HLA compatibility, with a preference for MSD or matched unrelated donors (MUD). The donors were primarily MUDs, as only one of our patients had a MSD.
Regarding engraftment, our patients engrafted relatively early for neutrophils, that occurred on day +15 on average. Only one of our patients didn’t successfully engraft, and she passed away at day +31 as detailed above. None of our patients experienced GVHD. In literature review, engraftment success rates are generally high, ranging from 80-90%, though delayed engraftment was more common in patients with active MDRO infections3,7,8. Acute GVHD (grade II-IV) occurred in 20-40% of patients, while chronic GVHD affected 10-20%7,8. Prophylactic strategies included PTCy, calcineurin inhibitors, and methotrexate5,7.
Regional data from the Middle East and Asia have reported comparable challenges in managing severe infections in patients with aplastic anemia undergoing HSCT6,10. Studies have demonstrated that reduced-intensity conditioning combined with meticulous infection control measures can achieve survival rates comparable to international cohorts. For example, Alasbali et al. reported favorable outcomes among SAA patients undergoing HSCT using fludarabine-based conditioning regimens in Saudi transplant centers6. Similarly, in there clinical practice guidelines for comprehensive management of allogeneic HSCT in patients with SAA, Xu et al highlighted the increasing prevalence of multidrug-resistant Gram-negative infections in hematopoietic transplant recipients, emphasizing the importance of early infection stabilization and individualized antimicrobial strategies prior to transplantation10. These findings support the feasibility of HSCT in patients with complex infectious profiles when multidisciplinary management strategies are implemented.
Overall survival rates post-transplant ranges from 60-80%, with infection-related mortality being a significant concern4. Early post-transplant period poses the highest risk for severe infections and sepsis3. Long-term survival is achievable in patients who were successfully managed for infections and avoided severe GVHD7,8. High engraftment rates and acceptable long-term survival highlight the potential of BMT even in the presence of MDROs, though the risk of severe infections remains substantial.
Conclusion
This case series demonstrates that hematopoietic stem cell transplantation (HSCT) can be successfully performed in pediatric patients with severe aplastic anemia (SAA) complicated by multidrug-resistant organism (MDRO) infections when meticulous infection control and multidisciplinary management are implemented. Despite the traditionally high risk associated with MDRO colonization or infection, our experience shows that early HSCT, guided by individualized antimicrobial strategies and close collaboration between infectious disease and transplant teams, can achieve favorable outcomes.
The key determinants of success included early infection stabilization, aggressive infection surveillance, and prompt neutrophil engraftment. These factors collectively facilitated immune recovery and minimized infection-related complications. Although one patient in our series succumbed to carbapenem-resistant Klebsiella pneumoniae sepsis, the remaining patients demonstrated sustained donor chimerism and long-term infection-free survival.
Our findings support the position that active or recent MDRO infection should not constitute an absolute contraindication to proceeding with HSCT in pediatric SAA. Timely transplantation, combined with rigorous infection control, can offer a curative outcome for these high-risk patients. Future multicenter studies are needed to develop standardized guidelines for pre-transplant infection stabilization, peri-transplant antimicrobial management, and post-transplant infection surveillance in this vulnerable population.
Conflict of interest: There are no conflicts of interest to declare from the authors.
The study was conducted in accordance with ethical guidelines and that all necessary consents were secured from the caregivers of the patients.
License
© The Author(s) 2026.
This is an open access article distributed under the terms of the Creative Commons Attribution 4.0 International License (CC BY 4.0), which permits unrestricted use, distribution, and reproduction in any medium, and unrestricted adaptation and reuse, including for commercial purposes, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
To view a copy of this license, visit https://creativecommons.org/licenses/by/4.0/.
References
-
Jamil A, Mukkamalla SKR. Lymphoma. StatPearls. 2023 Jul 1
-
Liu W, Liu J, Song Y, Wang X, Mi L, Cai C, et al. Burden of lymphoma in China, 1990-2019: an analysis of the global burden of diseases, injuries, and risk factors study 2019. Aging (Albany NY). 2022 Apr 4;14(7):3175.
-
Aussedat G, Traverse-Glehen A, Stamatoullas A, Molina T, Safar V, Laurent C, et al. Composite and sequential lymphoma between classical Hodgkin lymphoma and primary mediastinal lymphoma/diffuse large B-cell lymphoma, a clinico-pathological series of 25 cases. Br J Haematol. 2020 Apr 1;189(2):244-56.
-
Eyre TA, Gatter K, Collins GP, Hall GW, Watson C, Hatton CSR. Incidence, management, and outcome of high-grade transformation of nodular lymphocyte predominant Hodgkin lymphoma: long-term outcomes from a 30-year experience. Am J Hematol. 2015 Jun 1;90(6):E103-10.
-
Tao Y, Chen H, Liu D, Dai X. Survival among patients with composite and sequential lymphoma between primary mediastinal lymphoma/diffuse large B-cell lymphoma and classical Hodgkin lymphoma: a population-based study. Leuk Res. 2021;111.
-
Balodis A, Pimenova DA, Nikulshin SF, Balode G, Hasnere S. Rare case of Hodgkin lymphoma transformation into diffuse large B-cell lymphoma with atypical spread epidurally, intradurally and intramedullary: a case report. Am J Case Rep. 2022;23(1):e935014-1.
-
Thorn CF, Oshiro C, Marsh S, Hernandez-Boussard T, McLeod H, Klein TE, et al. Doxorubicin pathways: pharmacodynamics and adverse effects. Pharmacogenet Genomics. 2011;21(7):440
-
Henriksen PA. Anthracycline cardiotoxicity: an update on mechanisms, monitoring and prevention. Heart. 2018 Jun 1;104(12):971-7.
-
Aussedat G, Traverse-Glehen A, Stamatoullas A, Molina T, Safar V, Laurent C, et al. Composite and sequential lymphoma between classical Hodgkin lymphoma and primary mediastinal lymphoma/diffuse large B-cell lymphoma, a clinico-pathological series of 25 cases. Br J Haematol. 2020 Apr 1;189(2):244-56.
-
Chu DT, Nguyen TT, Tien NLB, Tran DK, Jeong JH, Anh PG, et al. Recent progress of stem cell therapy in cancer treatment: molecular mechanisms and potential applications. Cells. 2020 Feb 28;9(3).





